OMNIA SCIENTIA
OMNIA SCIENTIA
OMNIA SCIENTIA
OMNIA SCIENTIA
OMNIA SCIENTIA
PHYSICS & CHEMISTRY
PHYSICS & CHEMISTRY
PHYSICS & CHEMISTRY
PHYSICS & CHEMISTRY
PHYSICS & CHEMISTRY
COULOMB’S LAW
COULOMB’S LAW
COULOMB’S LAW
COULOMB’S LAW
COULOMB’S LAW
FORMULA & CALCULATOR
FORMULA & CALCULATOR
FORMULA & CALCULATOR
FORMULA & CALCULATOR
FORMULA & CALCULATOR
FORMULA & CALCULATOR
FORMULA & CALCULATOR
FORMULA & CALCULATOR
FORMULA & CALCULATOR
 |
 |
 |
 |
 |
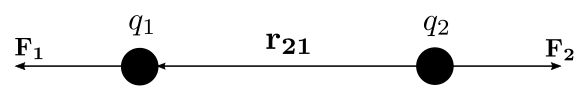
| The Coulomb or Coulomb’s inverse-square law describes the electrostatic interaction between electrically charged particles. Analogous to the inverse-square law of universal gravitation by Isaac Newton and is used to derive Gauss’s law, and vice versa. This law states: The magnitude of the electrostatic force of interaction between two point charges is directly proportional to the scalar multiplication of the magnitudes of charges and inversely proportional to the square of the distance between them. The force is along the straight line joining them. If the two charges have the same sign, the electrostatic force between them is repulsive; if they have different signs, the force between them is attractive. Coulomb’s law can also be stated as a simple mathematical expression. The scalar and vector forms of the mathematical equations are: |
| The Coulomb or Coulomb’s inverse-square law describes the electrostatic interaction between electrically charged particles. Analogous to the inverse-square law of universal gravitation by Isaac Newton and is used to derive Gauss’s law, and vice versa. This law states: The magnitude of the electrostatic force of interaction between two point charges is directly proportional to the scalar multiplication of the magnitudes of charges and inversely proportional to the square of the distance between them. The force is along the straight line joining them. If the two charges have the same sign, the electrostatic force between them is repulsive; if they have different signs, the force between them is attractive. Coulomb’s law can also be stated as a simple mathematical expression. The scalar and vector forms of the mathematical equations are: |
| The Coulomb or Coulomb’s inverse-square law describes the electrostatic interaction between electrically charged particles. Analogous to the inverse-square law of universal gravitation by Isaac Newton and is used to derive Gauss’s law, and vice versa. This law states: The magnitude of the electrostatic force of interaction between two point charges is directly proportional to the scalar multiplication of the magnitudes of charges and inversely proportional to the square of the distance between them. The force is along the straight line joining them. If the two charges have the same sign, the electrostatic force between them is repulsive; if they have different signs, the force between them is attractive. Coulomb’s law can also be stated as a simple mathematical expression. The scalar and vector forms of the mathematical equations are: |
| The Coulomb or Coulomb’s inverse-square law describes the electrostatic interaction between electrically charged particles. Analogous to the inverse-square law of universal gravitation by Isaac Newton and is used to derive Gauss’s law, and vice versa. This law states: The magnitude of the electrostatic force of interaction between two point charges is directly proportional to the scalar multiplication of the magnitudes of charges and inversely proportional to the square of the distance between them. The force is along the straight line joining them. If the two charges have the same sign, the electrostatic force between them is repulsive; if they have different signs, the force between them is attractive. Coulomb’s law can also be stated as a simple mathematical expression. The scalar and vector forms of the mathematical equations are: |
| The Coulomb or Coulomb’s inverse-square law describes the electrostatic interaction between electrically charged particles. Analogous to the inverse-square law of universal gravitation by Isaac Newton and is used to derive Gauss’s law, and vice versa. This law states: The magnitude of the electrostatic force of interaction between two point charges is directly proportional to the scalar multiplication of the magnitudes of charges and inversely proportional to the square of the distance between them. The force is along the straight line joining them. If the two charges have the same sign, the electrostatic force between them is repulsive; if they have different signs, the force between them is attractive. Coulomb’s law can also be stated as a simple mathematical expression. The scalar and vector forms of the mathematical equations are: |
| $|F|={ k }_{ e }\frac { |q1q2| }{ { r }^{ 2 } }$ | ${ F }_{ 1 }={ k }_{ e }\frac { q1q2 }{ { |{ { r }_{ 21 } }| }^{ 2 } } { \hat { r } }_{ 21 }$ |
| $|F|={ k }_{ e }\frac { |q1q2| }{ { r }^{ 2 } }$ | ${ F }_{ 1 }={ k }_{ e }\frac { q1q2 }{ { |{ { r }_{ 21 } }| }^{ 2 } } { \hat { r } }_{ 21 }$ |
| $|F|={ k }_{ e }\frac { |q1q2| }{ { r }^{ 2 } }$ | ${ F }_{ 1 }={ k }_{ e }\frac { q1q2 }{ { |{ { r }_{ 21 } }| }^{ 2 } } { \hat { r } }_{ 21 }$ |
| $|F|={ k }_{ e }\frac { |q1q2| }{ { r }^{ 2 } }$ | ${ F }_{ 1 }={ k }_{ e }\frac { q1q2 }{ { |{ { r }_{ 21 } }| }^{ 2 } } { \hat { r } }_{ 21 }$ |
| $|F|={ k }_{ e }\frac { |q1q2| }{ { r }^{ 2 } }$ | ${ F }_{ 1 }={ k }_{ e }\frac { q1q2 }{ { |{ { r }_{ 21 } }| }^{ 2 } } { \hat { r } }_{ 21 }$ |
| where the Coulomb’s constant (${ k }_{ e }=8.987’551’787’368’176’4$ x ${ 10 }^{ 9 } N\cdot { m }^{ 2 }\cdot { C }^{ -2 }$), $q1$ and $q2$ are the signed magnitudes of the charges, the scalar $r$ is the distance between the charges, the vector ${ r }_{ 21 }={ r }_{ 1 }-{ r }_{ 2 }$ is the vectorial distance between the charges, and ${ \hat { r } }_{ 21 }={ r }_{ 21 }/|{ { r }_{ 21 } }|$ (a vector unit vector pointing from $q2$ to $q1$). The vector form of the equation calculates the force ${ F }_{ 1 }$ applied on $q1$ by $q2$. If ${ r }_{ 21 }$ is used instead, then the effect on $q2$ can be found. It can be also calculated using Newton’s third law: ${ F }_{ 2 }={ F }_{ 1 }$. |
| where the Coulomb’s constant (${ k }_{ e }=8.987’551’787’368’176’4$ x ${ 10 }^{ 9 } N\cdot { m }^{ 2 }\cdot { C }^{ -2 }$), $q1$ and $q2$ are the signed magnitudes of the charges, the scalar $r$ is the distance between the charges, the vector ${ r }_{ 21 }={ r }_{ 1 }-{ r }_{ 2 }$ is the vectorial distance between the charges, and ${ \hat { r } }_{ 21 }={ r }_{ 21 }/|{ { r }_{ 21 } }|$ (a vector unit vector pointing from $q2$ to $q1$). The vector form of the equation calculates the force ${ F }_{ 1 }$ applied on $q1$ by $q2$. If ${ r }_{ 21 }$ is used instead, then the effect on $q2$ can be found. It can be also calculated using Newton’s third law: ${ F }_{ 2 }={ F }_{ 1 }$. |
| where the Coulomb’s constant (${ k }_{ e }=8.987’551’787’368’176’4$ x ${ 10 }^{ 9 } N\cdot { m }^{ 2 }\cdot { C }^{ -2 }$), $q1$ and $q2$ are the signed magnitudes of the charges, the scalar $r$ is the distance between the charges, the vector ${ r }_{ 21 }={ r }_{ 1 }-{ r }_{ 2 }$ is the vectorial distance between the charges, and ${ \hat { r } }_{ 21 }={ r }_{ 21 }/|{ { r }_{ 21 } }|$ (a vector unit vector pointing from $q2$ to $q1$). The vector form of the equation calculates the force ${ F }_{ 1 }$ applied on $q1$ by $q2$. If ${ r }_{ 21 }$ is used instead, then the effect on $q2$ can be found. It can be also calculated using Newton’s third law: ${ F }_{ 2 }={ F }_{ 1 }$. |
| where the Coulomb’s constant (${ k }_{ e }=8.987’551’787’368’176’4$ x ${ 10 }^{ 9 } N\cdot { m }^{ 2 }\cdot { C }^{ -2 }$), $q1$ and $q2$ are the signed magnitudes of the charges, the scalar $r$ is the distance between the charges, the vector ${ r }_{ 21 }={ r }_{ 1 }-{ r }_{ 2 }$ is the vectorial distance between the charges, and ${ \hat { r } }_{ 21 }={ r }_{ 21 }/|{ { r }_{ 21 } }|$ (a vector unit vector pointing from $q2$ to $q1$). The vector form of the equation calculates the force ${ F }_{ 1 }$ applied on $q1$ by $q2$. If ${ r }_{ 21 }$ is used instead, then the effect on $q2$ can be found. It can be also calculated using Newton’s third law: ${ F }_{ 2 }={ F }_{ 1 }$. |
| where the Coulomb’s constant (${ k }_{ e }=8.987’551’787’368’176’4$ x ${ 10 }^{ 9 } N\cdot { m }^{ 2 }\cdot { C }^{ -2 }$), $q1$ and $q2$ are the signed magnitudes of the charges, the scalar $r$ is the distance between the charges, the vector ${ r }_{ 21 }={ r }_{ 1 }-{ r }_{ 2 }$ is the vectorial distance between the charges, and ${ \hat { r } }_{ 21 }={ r }_{ 21 }/|{ { r }_{ 21 } }|$ (a vector unit vector pointing from $q2$ to $q1$). The vector form of the equation calculates the force ${ F }_{ 1 }$ applied on $q1$ by $q2$. If ${ r }_{ 21 }$ is used instead, then the effect on $q2$ can be found. It can be also calculated using Newton’s third law: ${ F }_{ 2 }={ F }_{ 1 }$. |
| Discovered in 1784 by Charles Augustin de Coulomb |
| Discovered in 1784 by Charles Augustin de Coulomb |
| Discovered in 1784 by Charles Augustin de Coulomb |
| Discovered in 1784 by Charles Augustin de Coulomb |
| Discovered in 1784 by Charles Augustin de Coulomb |
