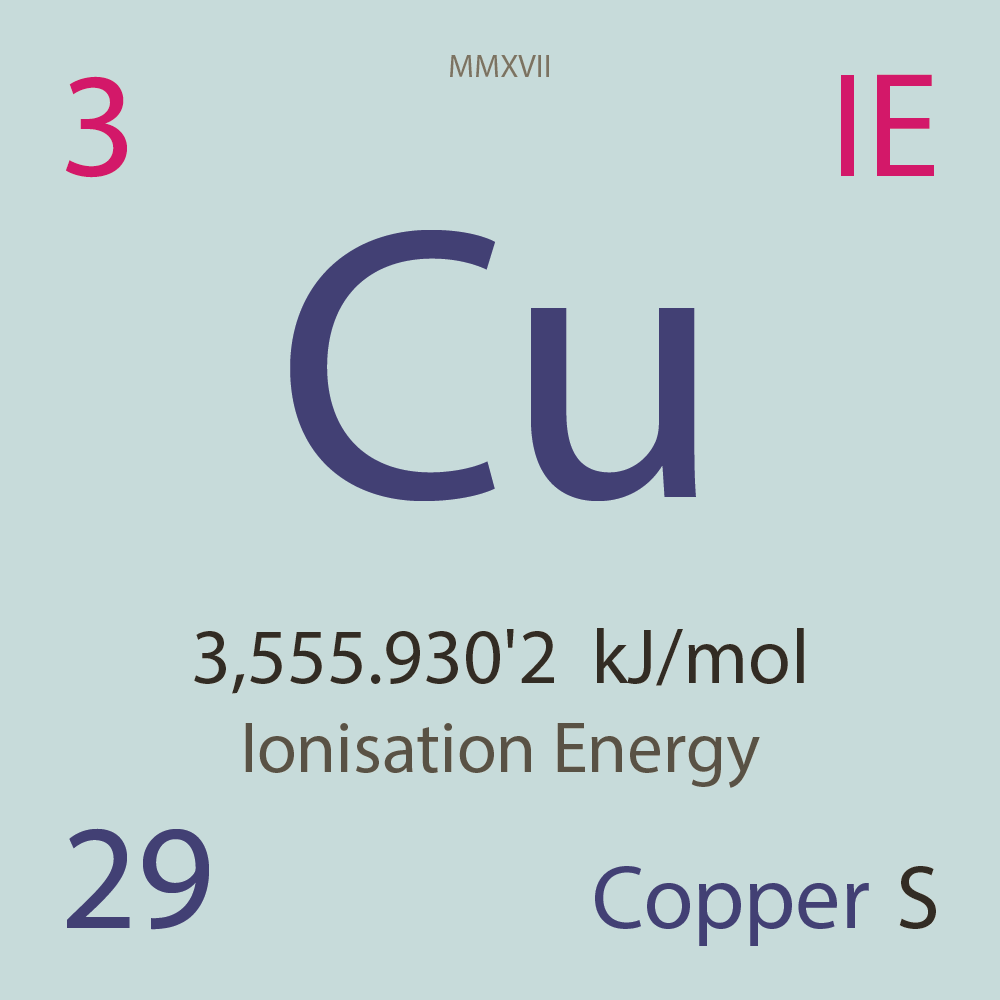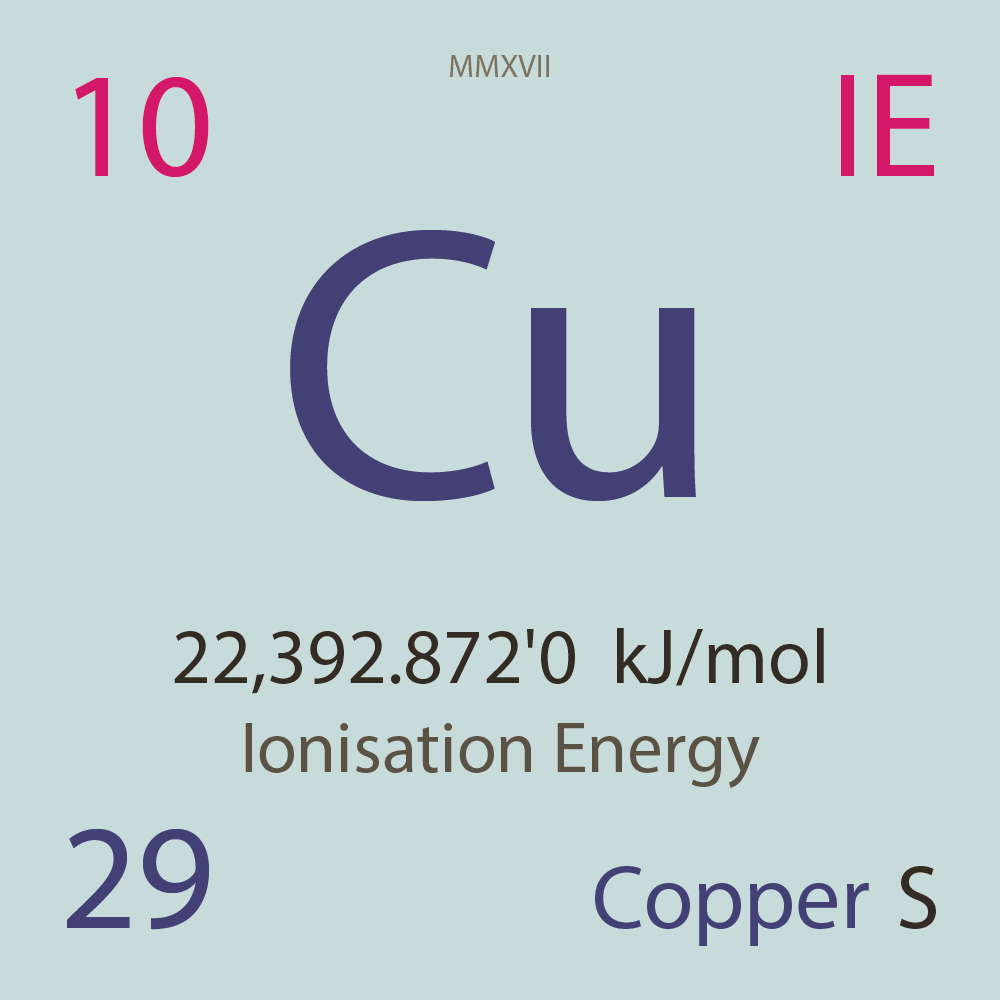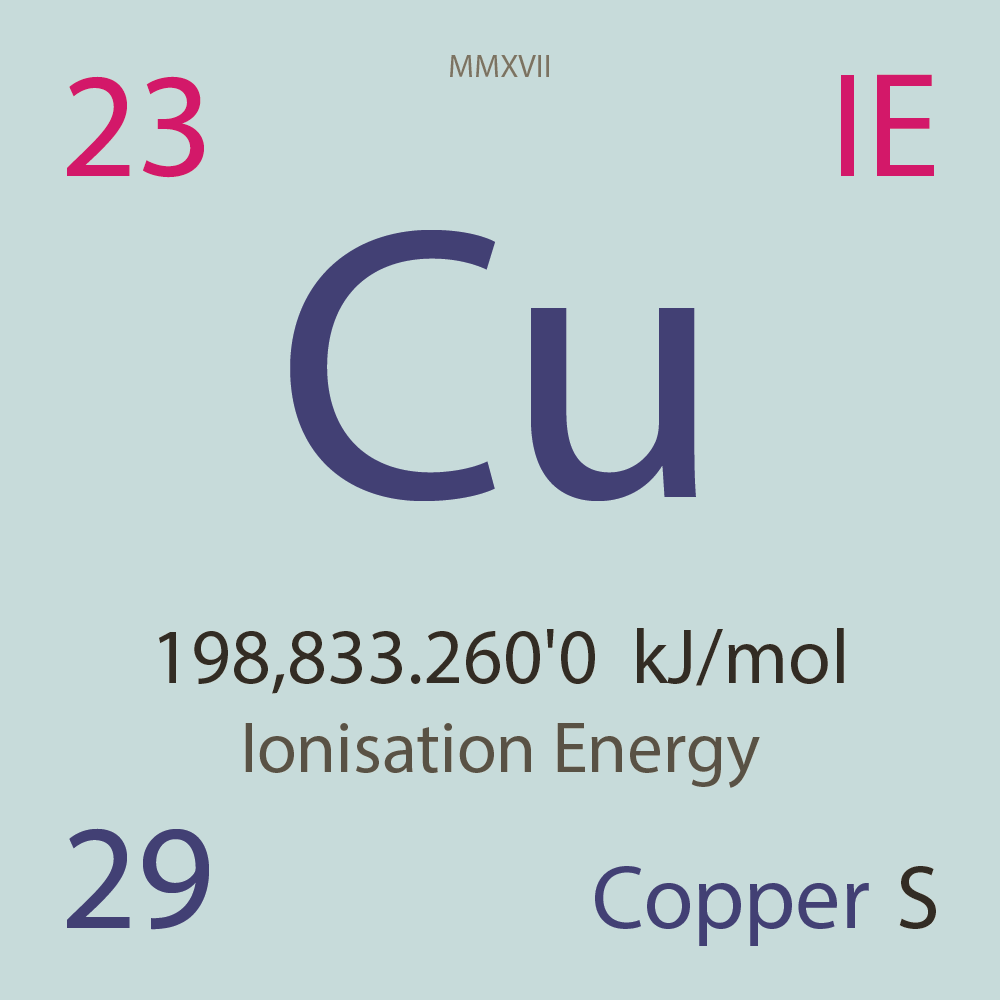| Isotope_029_cu_52_u |
Unstable |
⁵²Cu |
Fermion |
29 |
p |
23 |
n |
3 |
1 |
51.997'180'000'0 |
u |
~ 0 |
% |
~ 0 |
-2.627'000'000'0 |
MeV |
7.686'000'000'0 |
MeV |
- |
|
- |
|
? |
|
|
|
? |
% |
p |
1,523.000 |
keV |
⁵¹Ni |
⁵²Cu > [ ? % , p , 1,523.0 keV ] > ⁵¹Ni |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
? |
% |
⁵¹V |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Isotope_029_cu_53_u |
Unstable |
⁵³Cu |
Boson |
29 |
p |
24 |
n |
3/2 |
-1 |
52.985'550'000'0 |
u |
~ 0 |
% |
~ 0 |
-13.460'000'000'0 |
MeV |
7.897'000'000'0 |
MeV |
- |
|
- |
|
9.51E-15 |
year |
300.000 |
nano-seconds ( x⁻⁹ ) |
? |
% |
p |
1,905.000 |
keV |
⁵¹Ni |
⁵³Cu > [ ? % , p , 1,905.0 keV ] > ⁵¹Ni |
? |
% |
β+ |
14,888.000 |
keV |
⁵³Ni |
⁵³Cu > [ ? % , β+ , 14,888.0 keV ] > ⁵³Ni |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
? |
% |
⁵¹V |
? |
% |
⁵³Cr |
? |
% |
⁵²Cr |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Isotope_029_cu_54_u |
Unstable |
⁵⁴Cu |
Fermion |
29 |
p |
25 |
n |
3 |
1 |
53.976'710'000'0 |
u |
~ 0 |
% |
~ 0 |
-21.694'000'000'0 |
MeV |
8.053'000'000'0 |
MeV |
- |
|
- |
|
2.38E-15 |
year |
75.000 |
nano-seconds ( x⁻⁹ ) |
? |
% |
p |
387.000 |
keV |
⁵³Ni |
⁵⁴Cu > [ ? % , p , 387.0 keV ] > ⁵³Ni |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
? |
% |
⁵³Cr |
? |
% |
⁵²Cr |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Isotope_029_cu_55_u |
Unstable |
⁵⁵Cu |
Boson |
29 |
p |
26 |
n |
3/2 |
-1 |
54.966'050'000'0 |
u |
~ 0 |
% |
~ 0 |
-31.624'000'000'0 |
MeV |
8.234'000'000'0 |
MeV |
- |
|
- |
|
1.27E-9 |
year |
40.000 |
milli-seconds ( x⁻³ ) |
? |
% |
β+ |
12,689.000 |
keV |
⁵⁵Ni |
⁵⁵Cu > [ ? % , β+ , 12,689.0 keV ] > ⁵⁵Ni |
? |
% |
p |
298.000 |
keV |
⁵⁴Ni |
⁵⁵Cu > [ ? % , p , 298.0 keV ] > ⁵⁴Ni |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
? |
% |
⁵⁵Mn |
? |
% |
⁵⁴Cr |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Isotope_029_cu_56_u |
Unstable |
⁵⁶Cu |
Fermion |
29 |
p |
27 |
n |
4 |
1 |
55.958'560'000'0 |
u |
~ 0 |
% |
~ 0 |
-38.601'000'000'0 |
MeV |
8.355'000'000'0 |
MeV |
- |
|
- |
|
2.95E-9 |
year |
93.000 |
milli-seconds ( x⁻³ ) |
100.000'000 |
% |
β+ |
14,281.000 |
keV |
⁵⁶Ni |
⁵⁶Cu > [ 100 % , β+ , 14,281.0 keV ] > ⁵⁶Ni |
0.400'000 |
% |
β+p |
? |
keV |
⁵⁵Co |
⁵⁶Cu > [ 0.4 % , β+p , ? keV ] > ⁵⁵Co |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
100.000'000 |
% |
⁵⁶Fe |
0.400'000 |
% |
⁵⁵Mn |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Isotope_029_cu_57_u |
Unstable |
⁵⁷Cu |
Boson |
29 |
p |
28 |
n |
3/2 |
-1 |
56.949'211'078'0 |
u |
~ 0 |
% |
~ 0 |
-47.309'576'000'0 |
MeV |
8.503'274'000'0 |
MeV |
- |
|
- |
|
6.22E-9 |
year |
196.300 |
milli-seconds ( x⁻³ ) |
100.000'000 |
% |
β+ |
7,750.200 |
keV |
⁵⁷Ni |
⁵⁷Cu > [ 100 % , β+ , 7,750.2 keV ] > ⁵⁷Ni |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
100.000'000 |
% |
⁵⁷Fe |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Isotope_029_cu_58_u |
Unstable |
⁵⁸Cu |
Fermion |
29 |
p |
29 |
n |
1 |
1 |
57.944'538'499'0 |
u |
~ 0 |
% |
~ 0 |
-51.662'055'000'0 |
MeV |
8.570'869'000'0 |
MeV |
- |
|
- |
|
1.02E-7 |
year |
3.204 |
seconds ( x⁰ ) |
100.000'000 |
% |
β+ |
7,543.440 |
keV |
⁵⁸Ni |
⁵⁸Cu > [ 100 % , β+ , 7,543.44 keV ] > ⁵⁸Ni |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
? |
% |
⁵⁸Fe |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Isotope_029_cu_59_u |
Unstable |
⁵⁹Cu |
Boson |
29 |
p |
30 |
n |
3/2 |
-1 |
58.939'498'028'0 |
u |
~ 0 |
% |
~ 0 |
-56.357'224'000'0 |
MeV |
8.641'981'000'0 |
MeV |
- |
|
- |
|
2.58E-6 |
year |
81.480 |
seconds ( x⁰ ) |
100.000'000 |
% |
β+ |
3,776.227 |
keV |
⁵⁹Ni |
⁵⁹Cu > [ 100 % , β+ , 3,776.227 keV ] > ⁵⁹Ni |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
100.000'000 |
% |
⁵⁹Co |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Isotope_029_cu_60_u |
Unstable |
⁶⁰Cu |
Fermion |
29 |
p |
31 |
n |
2 |
1 |
59.937'365'030'0 |
u |
~ 0 |
% |
~ 0 |
-58.344'099'000'0 |
MeV |
8.665'585'000'0 |
MeV |
1.219'000'000'0 |
nm |
- |
|
4.50E-5 |
year |
1.420 |
kilo-seconds ( x³ ) |
100.000'000 |
% |
β+ |
5,105.780 |
keV |
⁵⁰Ni |
⁶⁰Cu > [ 100 % , β+ , 5,105.78 keV ] > ⁵⁰Ni |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
100.000'000 |
% |
⁶⁰Ni |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Isotope_029_cu_61_u |
Unstable |
⁶¹Cu |
Boson |
29 |
p |
32 |
n |
3/2 |
-1 |
60.933'457'821'0 |
u |
~ 0 |
% |
~ 0 |
-61.983'640'000'0 |
MeV |
8.715'507'000'0 |
MeV |
2.140'000'000'0 |
nm |
- |
|
3.80E-4 |
year |
11.988 |
kilo-seconds ( x³ ) |
100.000'000 |
% |
β+ |
1,215.050 |
keV |
⁶¹Ni |
⁶¹Cu > [ 100 % , β+ , 1,215.05 keV ] > ⁶¹Ni |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
100.000'000 |
% |
⁶¹Ni |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Isotope_029_cu_62_u |
Unstable |
⁶²Cu |
Fermion |
29 |
p |
33 |
n |
1 |
1 |
61.932'583'745'0 |
u |
~ 0 |
% |
~ 0 |
-62.797'837'000'0 |
MeV |
8.718'249'000'0 |
MeV |
-0.380'000'000'0 |
nm |
- |
|
1.84E-5 |
year |
580.398 |
seconds ( x⁰ ) |
100.000'000 |
% |
β+ |
2,926.060 |
keV |
⁶²Ni |
⁶²Cu > [ 100 % , β+ , 2,926.06 keV ] > ⁶²Ni |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
100.000'000 |
% |
⁶²Ni |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Isotope_029_cu_63_s |
Stable |
⁶³Cu |
Boson |
29 |
p |
34 |
n |
3/2 |
-1 |
62.929'597'474'0 |
u |
69.170'000 |
% |
43.528'402'572'8 |
-65.579'531'000'0 |
MeV |
8.752'134'000'0 |
MeV |
2.223'290'000'0 |
nm |
-0.211'000'000'0 |
b |
- |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Isotope_029_cu_64_u |
Unstable |
⁶⁴Cu |
Fermion |
29 |
p |
35 |
n |
1 |
1 |
63.929'764'183'0 |
u |
~ 0 |
% |
~ 0 |
-65.424'243'000'0 |
MeV |
8.739'070'000'0 |
MeV |
-0.217'000'000'0 |
nm |
- |
|
1.45E-3 |
year |
45.684 |
kilo-seconds ( x³ ) |
61.000'000 |
% |
β+ |
652.834 |
keV |
⁶⁴Ni |
⁶⁴Cu > [ 61 % , β+ , 652.834 keV ] > ⁶⁴Ni |
39.000'000 |
% |
β- |
579.353 |
keV |
⁶⁴Zn |
⁶⁴Cu > [ 39 % , β- , 579.353 keV ] > ⁶⁴Zn |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
61.000'000 |
% |
⁶⁴Ni |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Isotope_029_cu_65_s |
Stable |
⁶⁵Cu |
Boson |
29 |
p |
36 |
n |
3/2 |
-1 |
64.927'789'485'0 |
u |
30.830'000 |
% |
20.017'237'498'2 |
-67.263'661'000'0 |
MeV |
8.757'096'000'0 |
MeV |
2.381'670'000'0 |
nm |
-0.195'000'000'0 |
b |
- |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Isotope_029_cu_66_u |
Unstable |
⁶⁶Cu |
Fermion |
29 |
p |
37 |
n |
1 |
1 |
65.928'868'813'0 |
u |
~ 0 |
% |
~ 0 |
-66.258'274'000'0 |
MeV |
8.731'472'000'0 |
MeV |
-0.282'000'000'0 |
nm |
- |
|
9.73E-6 |
year |
307.020 |
seconds ( x⁰ ) |
100.000'000 |
% |
β- |
2,641.150 |
keV |
⁶⁶Zn |
⁶⁶Cu > [ 100 % , β- , 2,641.15 keV ] > ⁶⁶Zn |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
100.000'000 |
% |
⁶⁶Zn |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Isotope_029_cu_67_u |
Unstable |
⁶⁷Cu |
Boson |
29 |
p |
38 |
n |
3/2 |
-1 |
66.927'730'314'0 |
u |
~ 0 |
% |
~ 0 |
-67.318'779'000'0 |
MeV |
8.737'447'000'0 |
MeV |
- |
|
- |
|
7.05E-3 |
year |
222.601 |
kilo-seconds ( x³ ) |
100.000'000 |
% |
β- |
561.660 |
keV |
⁶⁷Zn |
⁶⁷Cu > [ 100 % , β- , 561.66 keV ] > ⁶⁷Zn |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
100.000'000 |
% |
⁶⁷Zn |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Isotope_029_cu_68_u |
Unstable |
⁶⁸Cu |
Fermion |
29 |
p |
39 |
n |
1 |
1 |
67.929'610'889'0 |
u |
~ 0 |
% |
~ 0 |
-65.567'035'000'0 |
MeV |
8.701'890'000'0 |
MeV |
- |
|
- |
|
9.85E-7 |
year |
31.100 |
seconds ( x⁰ ) |
100.000'000 |
% |
β- |
4,440.190 |
keV |
⁶⁸Zn |
⁶⁸Cu > [ 100 % , β- , 4,440.19 keV ] > ⁶⁸Zn |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
100.000'000 |
% |
⁶⁸Zn |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Isotope_029_cu_69_u |
Unstable |
⁶⁹Cu |
Boson |
29 |
p |
40 |
n |
3/2 |
-1 |
68.929'429'269'0 |
u |
~ 0 |
% |
~ 0 |
-65.736'213'000'0 |
MeV |
8.695'203'000'0 |
MeV |
- |
|
- |
|
5.42E-6 |
year |
171.000 |
seconds ( x⁰ ) |
100.000'000 |
% |
β- |
2,681.760 |
keV |
⁶⁹Zn |
⁶⁹Cu > [ 100 % , β- , 2,681.76 keV ] > ⁶⁹Zn |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
100.000'000 |
% |
⁶⁹Ga |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Isotope_029_cu_70_u |
Unstable |
⁷⁰Cu |
Fermion |
29 |
p |
41 |
n |
6 |
-1 |
69.932'392'343'0 |
u |
~ 0 |
% |
~ 0 |
-62.976'127'000'0 |
MeV |
8.646'861'000'0 |
MeV |
- |
|
- |
|
1.41E-6 |
year |
44.500 |
seconds ( x⁰ ) |
100.000'000 |
% |
β- |
6,588.520 |
keV |
⁷⁰Zn |
⁷⁰Cu > [ 100 % , β- , 6,588.52 keV ] > ⁷⁰Zn |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
? |
% |
⁷⁰Ge |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Isotope_029_cu_71_u |
Unstable |
⁷¹Cu |
Boson |
29 |
p |
42 |
n |
3/2 |
-1 |
70.932'676'833'0 |
u |
~ 0 |
% |
~ 0 |
-62.711'127'000'0 |
MeV |
8.635'022'000'0 |
MeV |
- |
|
- |
|
6.15E-7 |
year |
19.400 |
seconds ( x⁰ ) |
100.000'000 |
% |
β- |
4,615.800 |
keV |
⁷¹Zn |
⁷¹Cu > [ 100 % , β- , 4,615.8 keV ] > ⁷¹Zn |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
100.000'000 |
% |
⁷¹Ge |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Isotope_029_cu_72_u |
Unstable |
⁷²Cu |
Fermion |
29 |
p |
43 |
n |
1 |
1 |
71.935'820'307'0 |
u |
~ 0 |
% |
~ 0 |
-59.782'999'000'0 |
MeV |
8.586'525'000'0 |
MeV |
- |
|
- |
|
2.09E-7 |
year |
6.600 |
seconds ( x⁰ ) |
100.000'000 |
% |
β- |
8,348.380 |
keV |
⁷²Zn |
⁷²Cu > [ 100 % , β- , 8,348.38 keV ] > ⁷²Zn |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
100.000'000 |
% |
⁷²Ge |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Isotope_029_cu_73_u |
Unstable |
⁷³Cu |
Boson |
29 |
p |
44 |
n |
3/2 |
-1 |
72.936'675'282'0 |
u |
~ 0 |
% |
~ 0 |
-58.986'595'000'0 |
MeV |
8.568'557'000'0 |
MeV |
- |
|
- |
|
1.33E-7 |
year |
4.200 |
seconds ( x⁰ ) |
100.000'000 |
% |
β- |
6,423.700 |
keV |
⁷³Zn |
⁷³Cu > [ 100 % , β- , 6,423.7 keV ] > ⁷³Zn |
? |
% |
β-n |
1,073.470 |
keV |
⁷²Zn |
⁷³Cu > [ ? % , β-n , 1,073.47 keV ] > ⁷²Zn |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
100.000'000 |
% |
⁷³Ge |
? |
% |
⁷²Ge |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Isotope_029_cu_74_u |
Unstable |
⁷⁴Cu |
Fermion |
29 |
p |
45 |
n |
1 |
1 |
73.939'874'862'0 |
u |
~ 0 |
% |
~ 0 |
-56.006'205'000'0 |
MeV |
8.521'562'000'0 |
MeV |
- |
|
- |
|
5.05E-8 |
year |
1.594 |
seconds ( x⁰ ) |
100.000'000 |
% |
β- |
9,702.700 |
keV |
⁷⁴Zn |
⁷⁴Cu > [ 100 % , β- , 9,702.7 keV ] > ⁷⁴Zn |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
100.000'000 |
% |
⁷⁴Ge |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Isotope_029_cu_75_u |
Unstable |
⁷⁵Cu |
Boson |
29 |
p |
46 |
n |
3/2 |
-1 |
74.941'900'000'0 |
u |
~ 0 |
% |
~ 0 |
-54.119'802'000'0 |
MeV |
8.490'407'000'0 |
MeV |
- |
|
- |
|
3.88E-8 |
year |
1.224 |
seconds ( x⁰ ) |
96.000'000 |
% |
β- |
8,349.000 |
keV |
⁷⁵Zn |
⁷⁵Cu > [ 96 % , β- , 8,349.0 keV ] > ⁷⁵Zn |
3.500'000 |
% |
β-n |
3,518.000 |
keV |
⁷⁴Zn |
⁷⁵Cu > [ 3.5 % , β-n , 3,518.0 keV ] > ⁷⁴Zn |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
96.000'000 |
% |
⁷⁵As |
3.500'000 |
% |
⁷⁴Ge |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Isotope_029_cu_76_u |
Unstable |
⁷⁶Cu |
Fermion |
29 |
p |
47 |
n |
? |
0 |
75.945'275'026'0 |
u |
~ 0 |
% |
~ 0 |
-50.975'985'000'0 |
MeV |
8.443'527'000'0 |
MeV |
- |
|
- |
|
2.03E-8 |
year |
641.000 |
milli-seconds ( x⁻³ ) |
97.000'000 |
% |
β- |
11,160.700 |
keV |
⁷⁶Zn |
⁷⁶Cu > [ 97 % , β- , 11,160.7 keV ] > ⁷⁶Zn |
3.000'000 |
% |
β-n |
3,421.700 |
keV |
⁷⁵Zn |
⁷⁶Cu > [ 3 % , β-n , 3,421.7 keV ] > ⁷⁵Zn |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
97.000'000 |
% |
⁷⁶Se |
3.000'000 |
% |
⁷⁵As |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Isotope_029_cu_77_u |
Unstable |
⁷⁷Cu |
Boson |
29 |
p |
48 |
n |
3/2 |
-1 |
76.947'850'000'0 |
u |
~ 0 |
% |
~ 0 |
-48.577'000'000'0 |
MeV |
8.408'000'000'0 |
MeV |
- |
|
- |
|
1.49E-8 |
year |
469.000 |
milli-seconds ( x⁻³ ) |
100.000'000 |
% |
β- |
10,145.000 |
keV |
⁷⁷Zn |
⁷⁷Cu > [ 100 % , β- , 10,145.0 keV ] > ⁷⁷Zn |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
100.000'000 |
% |
⁷⁷Se |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Isotope_029_cu_78_u |
Unstable |
⁷⁸Cu |
Fermion |
29 |
p |
49 |
n |
? |
0 |
77.951'960'000'0 |
u |
~ 0 |
% |
~ 0 |
-44.749'000'000'0 |
MeV |
8.354'000'000'0 |
MeV |
- |
|
- |
|
1.08E-8 |
year |
342.000 |
milli-seconds ( x⁻³ ) |
100.000'000 |
% |
β- |
12,594.000 |
keV |
⁷⁸Zn |
⁷⁸Cu > [ 100 % , β- , 12,594.0 keV ] > ⁷⁸Zn |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
100.000'000 |
% |
⁷⁸Se |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Isotope_029_cu_79_u |
Unstable |
⁷⁹Cu |
Boson |
29 |
p |
50 |
n |
3/2 |
-1 |
78.954'560'000'0 |
u |
~ 0 |
% |
~ 0 |
-42.327'000'000'0 |
MeV |
8.320'000'000'0 |
MeV |
- |
|
- |
|
5.96E-11 |
year |
1.880 |
milli-seconds ( x⁻³ ) |
55.000'000 |
% |
β-n |
6,944.000 |
keV |
⁷⁸Zn |
⁷⁹Cu > [ 55 % , β-n , 6,944.0 keV ] > ⁷⁸Zn |
45.000'000 |
% |
β- |
11,092.000 |
keV |
⁷⁹Zn |
⁷⁹Cu > [ 45 % , β- , 11,092.0 keV ] > ⁷⁹Zn |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
55.624'650 |
% |
⁷⁸Se |
44.550'000 |
% |
⁷⁹Br |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Isotope_029_cu_80_u |
Unstable |
⁸⁰Cu |
Fermion |
29 |
p |
51 |
n |
? |
0 |
79.960'870'000'0 |
u |
~ 0 |
% |
~ 0 |
-36.449'000'000'0 |
MeV |
8.243'000'000'0 |
MeV |
- |
|
- |
|
3.17E-9 |
year |
100.000 |
milli-seconds ( x⁻³ ) |
? |
% |
β- |
15,395.000 |
keV |
⁸⁰Zn |
⁸⁰Cu > [ ? % , β- , 15,395.0 keV ] > ⁸⁰Zn |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
? |
% |
⁷⁸Se |
? |
% |
⁷⁹Br |
? |
% |
⁸⁰Kr |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|